Owens, Wendt, Rabel and Kaelble (OWRK) method
The Owens, Wendt, Rabel and Kaelble method is a standard method for calculating the surface free energy of a solid from the contact angle with several liquids. In doing so, the surface free energy is divided into a polar part and a disperse part.
Background
According to Young’s equation, there is a relationship between the contact angle θ, the surface tension of the liquid σl, the interfacial tension σsl between liquid and solid and the surface free energy σs of the solid:

In order to be able to calculate the surface free energy from the contact angle, the second unknown variable σsl must be determined.
Building on the Fowkes method, the interfacial tension σsl is calculated based on the two surface tensions σs and σl and the similar interactions between the phases. These interactions are interpreted as the geometric mean of a disperse part σD and a polar part σP of the surface tension or surface free energy:

At least two liquids with known disperse and polar parts of the surface tension are required to determine the surface free energy of the solid, wherein at least one of the liquids must have a polar part > 0.
According to the two-component model, the interfacial tension depends on whether polar and disperse parts can form interactions with corresponding parts of the adjacent phase. For example, the interfacial tension with respect to water as the polar liquid is smaller when the solid is also polar. If on the other hand, the polar part of the solid is small, then the square root term (σpl ⋅ σps)1/2 assumes a low value. The polar interactions then only make a small contribution to reducing the interfacial tension; this corresponds to poor wetting and a high contact angle.
The following diagram symbolizes the different interactions by means of hands - only "matching" hands can link with one another.
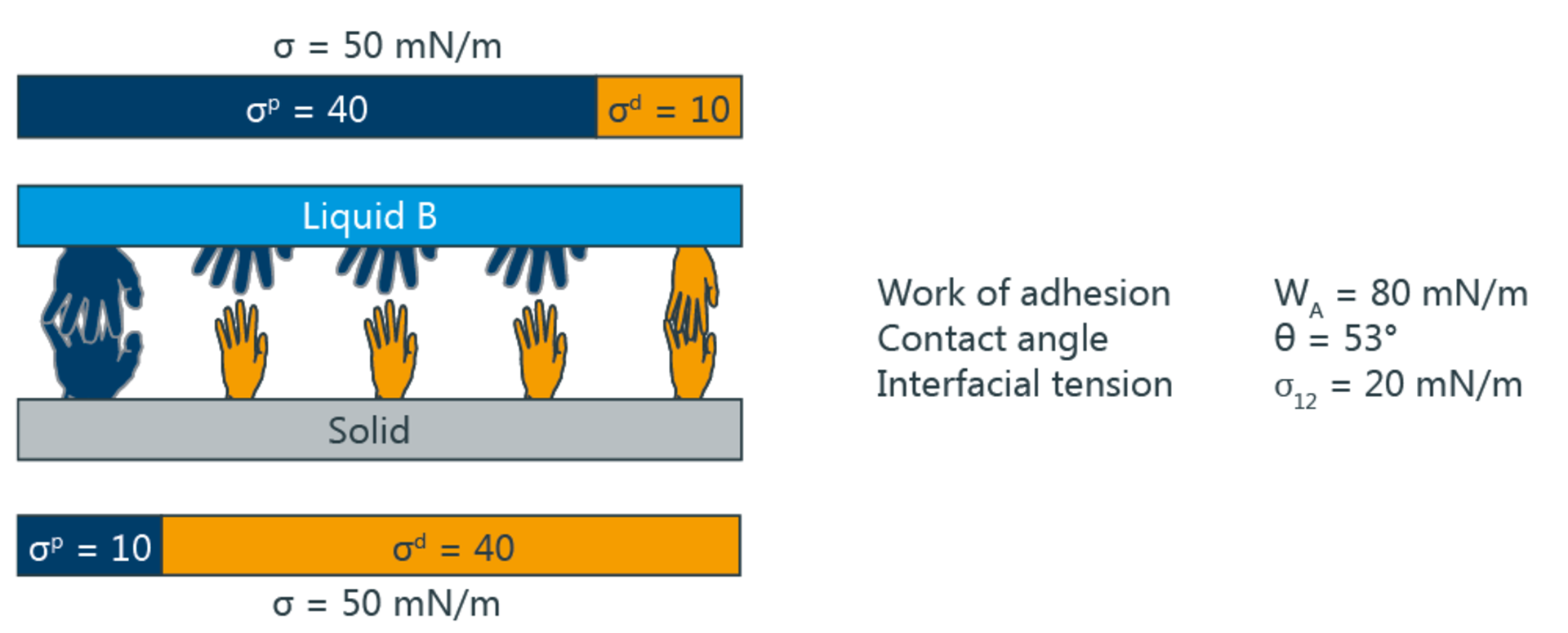
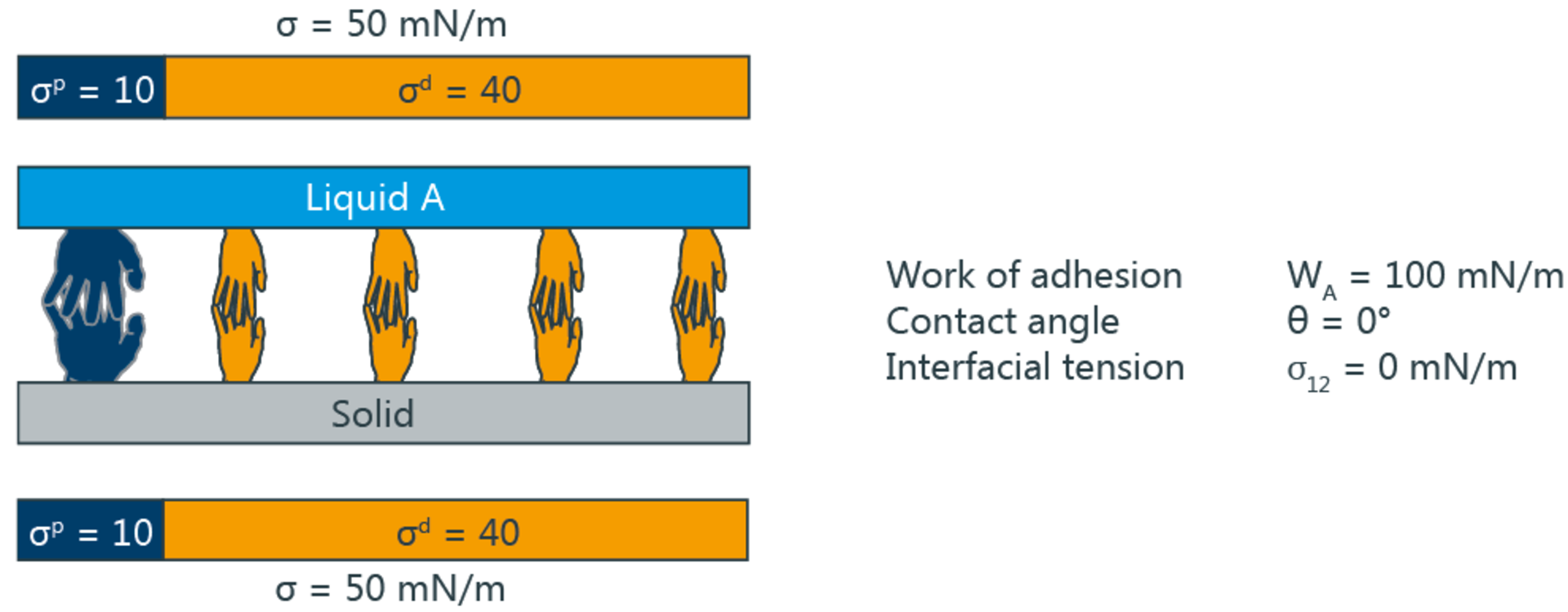
Significance
The OWRK method is used when investigating the effect of polar and disperse interactions on wettability and adhesion. In particular, the contact between surfaces of different polarity and the effect of change in polarity, for example by coating or plasma treatment, can be assessed and optimized with the help of the OWRK method - in processes such as painting, printing, bonding and hydrophobic or hydrophilic coating.
Bibliography
- D. H. Kaelble, Dispersion-Polar Surface Tension Properties of Organic Solids. In: J. Adhesion 2 (1970), P. 66-81.
- D. Owens; R. Wendt, Estimation of the Surface Free Energy of Polymers. In: J. Appl. Polym. Sci 13 (1969), P. 1741-1747.
- W. Rabel, Einige Aspekte der Benetzungstheorie und ihre Anwendung auf die Untersuchung und Veränderung der Oberflächeneigenschaften von Polymeren. In: Farbe und Lack 77,10 (1971), P. 997-1005.





