Emulsion
Emulsions are mixtures of two liquids not soluble in each other, in which droplets (disperse phase) are distributed in a surrounding phase (continuous phase) without visible inhomogeneity. Emulsions therefore belong to the group of mixtures known as dispersions, which are also called colloids. As a rule, an emulsion is a mixture of a hydrophobic liquid and an aqueous phase. Well-known examples of emulsions are milk and mayonnaise.
How emulsions are formed
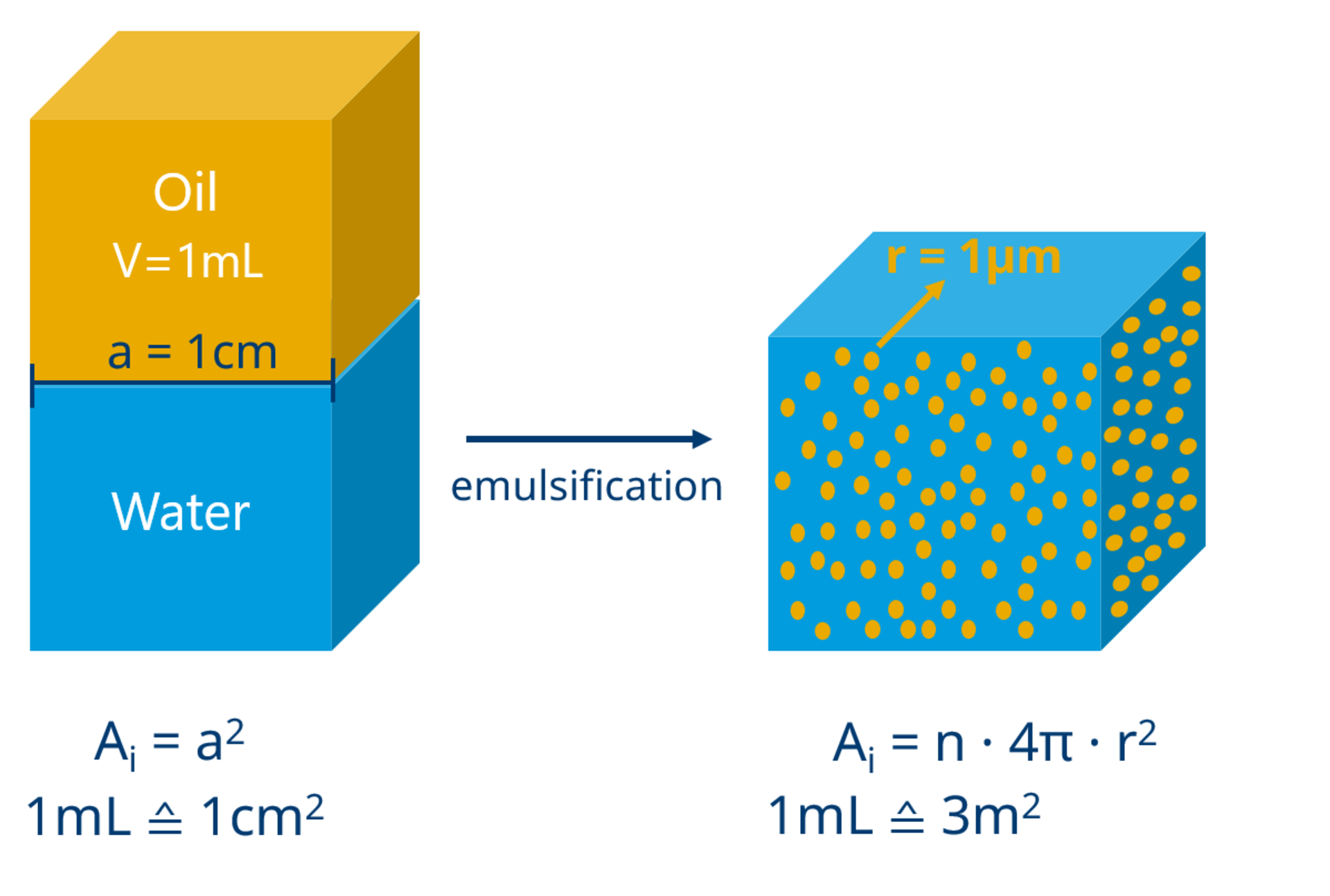
Since oil and water quickly separate again after mixing, additives known as emulsifiers are required (or are already naturally present) to form an emulsion. Emulsifiers are surfactants, i.e., substances that have an affinity for both water and oil (amphiphilic) and are adsorbed at the phase boundaries between the droplets and the continuous phase.
Surfactants reduce the interfacial tension between oil and water, which is equivalent to the work required to enlarge an interface. The division of a given volume into droplets leads to a larger interface; therefore, this process is facilitated by surfactants. The effort required for emulsification (e.g., by shaking or stirring) is reduced and phase separation is delayed.
What types of emulsions are there?
Surfactant-stabilized emulsions between oil and water
Regardless of the actual composition of the hydrophobic and aqueous phases, we generally refer to oil and water. Depending on which liquid forms the dispersed phase and which forms the continuous phase, a distinction is made between oil-in-water and water-in-oil emulsions (O/W or W/O). The volume fraction of the dispersed phase is decisive. If this volume fraction is increased, a critical point will be reached at which one emulsion type transforms into the other, a process known as phase inversion.

Microemulsions
With the help of suitable surfactant formulations, stable microemulsions with very small droplets can be formed, in which the interfacial tension against the continuous phase is almost 0 mN/m. Microemulsions are not considered true emulsions due to their completely different thermodynamic behavior.
▶ Find out more in our glossary article on the topic of microemulsions.
Pickering emulsions
Pickering emulsions are only partially stabilized by surfactants, if at all, but rather by solid particles that accumulate at the phase boundaries. These particles are generally wettable by water and oil and therefore have an amphiphilic function similar to surfactants. Pickering emulsions are often characterized by a very long shelf life and are used, for example, in the food, pharmaceutical, and cosmetics industries. Measurements of powder wettability based on the contact angle play an important role in the optimization of Pickering emulsions.
Are emulsions stable?
Although many emulsions have a long shelf life and appear unchanged, all true emulsions are subject to phase separation and are thermodynamically unstable. In many products, added stabilizers counteract this aging process, but usually do not stop it completely.
How does phase separation of emulsions occur and what role does interfacial tension play in this process?
Apart from possible chemical changes, various physical processes are responsible for the phase separation of an emulsion. The most important of these are essentially related to the interfacial tension between the dispersed and continuous phases.
- Coalescence: Due to the interfacial tension, the system strives for a smaller internal surface area with correspondingly larger droplets. Colliding droplets destroy the separating liquid film and flow together until the phases eventually separate more and more. The reduction of interfacial tension and the stabilization of the film by surfactants slow down this process.
- Ostwald ripening: Due to their greater curvature, smaller droplets have higher internal pressure. This pressure difference leads to a flow of matter towards the larger droplets. As a result, the large droplets grow, while the small ones eventually dissolve.
In this process, too, the speed depends on the interfacial tension, as this dominates the resulting curvature pressure (Laplace pressure). Emulsions with a homogeneous droplet size distribution undergo slower Ostwald ripening.
Ostwald ripening is a universal thermodynamic process in colloidal systems, which is also a driving factor in foam decay, for example. - Sedimentation: Due to the difference in density between the dispersed and continuous phases, droplets sink or rise ("creaming") over time, depending on which phase is the more specifically heavy one. This does not initially change the droplet size, but as the droplets get closer to each other, coalescence and Ostwald ripening—and thus interfacial tension—become more pronounced, ultimately leading to phase separation.
What are the benefits of measuring interfacial tension for the production of emulsions?
Interfacial tension (IFT) is not only crucial for the stability of emulsions, but also for the time and energy required for their production. This makes the IFT between the dispersed and continuous phases a key parameter that is of great importance in the development of new recipes and formulations as well as for quality control.
Tensiometry
The most common measurements in emulsion analysis are those taken with a force tensiometer, which measures the wetting force at a ring (Du Noüy ring method) or a plate (Wilhelmy plate method) at the phase boundary. Spinning drop tensiometers are also used and are particularly ideal for very low IFT values, such as those found in microemulsions. Alternatively, optical drop shape analysis can be used to measure the IFT based on the shape of a pendant drop in the surrounding phase.
▶ Our video shows you how to measure interfacial tension using the Wilhelmy plate method with the illustrative example of mixing a vinaigrette.
▶ For more information on the contribution of tensiometry to the development of emulsions, please refer to our application report AR291 ("Characterization of a Versatile Emulsifier for Low-Viscous Formulations and Liposomal Structures").
Interfacial rheology
Interfacial rheological measurements, which examine the change in IFT in response to the deformation of a drop in a two-phase system, are particularly informative. The resulting data on interfacial viscosity and elasticity allow conclusions to be drawn about emulsifiability and emulsion stability, as well as the behavior of emulsions in dynamic processes.
▶ Learn more in our application report AR285 ("Interfacial Rheology of Emulsifiers in Food").
What are typical examples and applications for emulsions?
Emulsions are found as consumer products in virtually every area of daily life and also play a role in many industrial processes:
- Food: In addition to predominantly natural emulsions such as milk, butter, or cream, there is a wide range of artificially produced emulsions. Examples include spreadable fats, ice cream, mustard, mayonnaise, vinaigrette, and other dressings, and more recently, vegan substitutes for dairy products...
- Cosmetics and personal care: Here, emulsions usually have the task of supplying the skin and hair with nourishing fats in a manner compatible with the body's hydrophilic environment: skin creams and sunscreen products, conditioners and hair rinses, liquid rouge...
- Pharmacy: In ointments, emulsions ensure even application to the skin and good absorption of the often hydrophobic active ingredients. Medicines for oral and intravenous administration can also be emulsions, as can certain nasal sprays.
- Enhanced oil recovery (EOR): In chemical flooding, surfactant mixtures are pumped into the reservoir, where they mobilize the oil adhering to the rock in the form of an emulsion. The aim is often to form a microemulsion.
- Chemical synthesis: Many polymers, including polystyrene and latex, are produced by emulsion polymerization. The reaction takes place in the droplets of the dispersed, non-aqueous phase.






