Contact angle
When an interface exists between a liquid and a solid, the angle between the surface of the liquid and the outline of the contact surface is described as the contact angle θ (lower case theta). The contact angle (wetting angle) is a measure of the wettability of a solid by a liquid.
Background
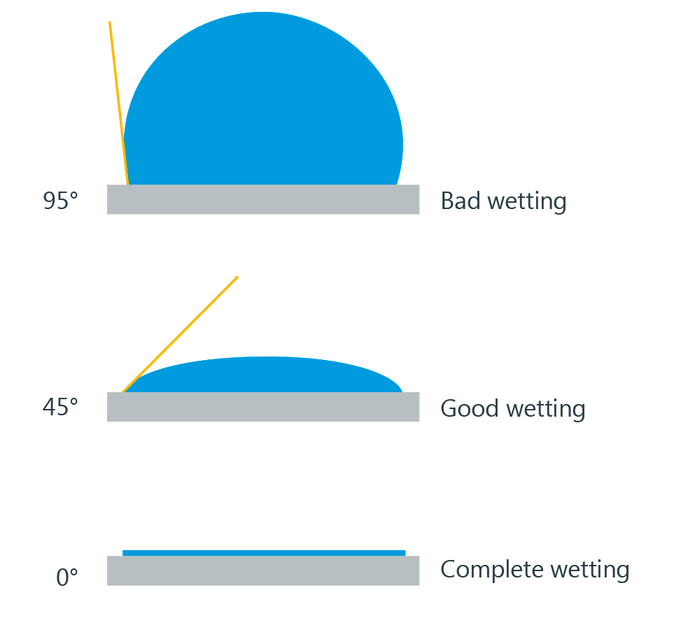
In the case of complete wetting (spreading), the contact angle is 0°. Between 0° and 90°, the solid is wettable and above 90° it is not wettable. In the case of ultrahydrophobic materials with the so-called lotus effect, the contact angle approaches the theoretical limit of 180°.
According to Young's equation, there is a relationship between the contact angle θ, the surface tension of the liquid σl, the interfacial tension σsl between liquid and solid and the surface free energy σs of the solid:

The contact angle with one or more liquids can be used to determine the surface free energy of a solid.
Significance
The contact angle is important wherever the intensity of the phase contact between liquid and solid substances needs to be checked or assessed: coating, painting, cleaning, printing, hydrophobic or hydrophilic coating, bonding, dispersing etc.
Measuring methods
- Drop shape analysis: The contact angle is measured using the image of a sessile drop at the points of intersection (three-phase contact points) between the drop contour and the projection of the surface (baseline).
- Wilhelmy plate method: The force acting in the tensile direction when moving a plate-shaped solid vertically in a liquid is measured. This force depends on the contact angle as well as on the surface tension and the wetted length.
- Powder contact angle measurement using the Washburn method: The increase in weight of an immersed powder-filled tube is measured with respect to time. The rate of rise of the liquid column depends, among other things, on the contact angle.
- Top-view distance method: The curvature of the drop surface associated with the contact angle is measured using the distance between light spots which are reflected on the top of a drop surface.






