Critical micelle concentration (CMC) and surfactant concentration
The CMC (critical micelle concentration) is the concentration of a surfactant in a bulk phase, above which aggregates of surfactant molecules, so-called micelles, start to form. The CMC is an important characteristic for surfactants.
Background
Surfactant molecules adsorb at the surface and reduce the surface tension (SFT). This decreases as the surfactant concentration in the solution increases. Once micelles form within a certain concentration range, the concentration of free surfactant molecules does not increase further and the surface tension remains approximately constant. This transition concentration is referred to as the CMC.

Significance
A knowledge of the CMC is very important when using surfactants. As the surface tension does not reduce further above the CMC, in many processes the CMC specifies the limiting concentration for meaningful use. When the formation of micelles is desirable, e.g. when cleaning, the CMC is a measure of the efficiency of a surfactant. Variables for the scientific characterisation of the surface adsorption can also be derived from the CMC. Examples include the area per molecule and the surface excess. The last-mentioned variable can be used to help determine the adsorption coefficient from measurements made with a bubble pressure tensiometer.
Measurement
The CMC is determined with a tensiometer by measuring the surface tension of a concentration series. With pure surfactants, the SFT is linearly dependent on the logarithm of the concentration over a large range. Above the CMC, the SFT is extensively independent of the concentration. The CMC results from the intersection between the regression straight line of the linearly dependent region and the straight line passing through the plateau.
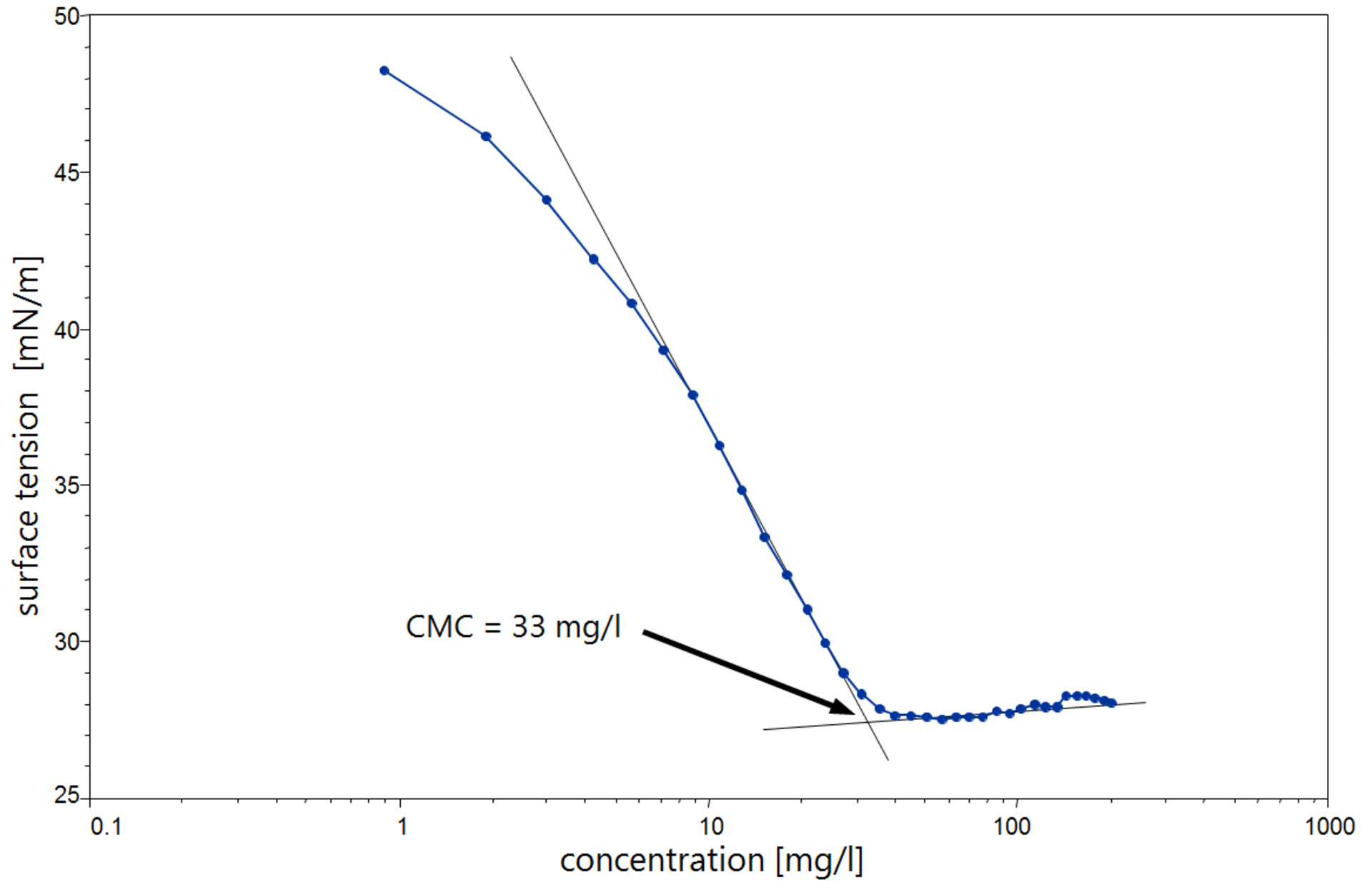
In modern tensiometers, the CMC is determined fully automatically using a dosing unit. The sample volume can be kept constant with the help of a second dosing unit which is used for extraction purposes. This enables a dilution series with very many concentrations to be obtained and therefore a very accurate determination of the CMC (extended CMC).
